Abstract
Introduction: Current guidelines support allogeneic hematopoietic cell transplantation (allo-HCT) in patients with acute leukemia who achieve complete remission (CR) and are at high risk for relapse. Patients with primary refractory disease or those refractory to re-induction therapy after relapse have a poor prognosis in the absence of allo-HCT. While several studies have addressed the role of allo-HCT in those settings, the measurable benefit of allo-HCT has not been defined in such patients. Most previous studies have included patients who received myeloablative conditioning and did not address the possible impact of molecular genetic risk factors.
Objective: The present study assessed outcomes of allo-HCT in patients with refractory or relapsed (R/R) acute leukemia receiving myeloablative (MAC) or reduced intensity conditioning (RIC) regimens. Our goal was to identify novel risk factors associated with improved outcomes to aid patient selection in the setting of R/R acute leukemia.
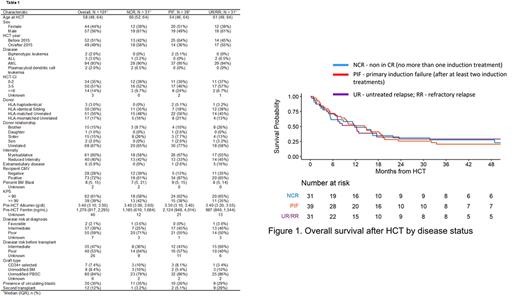
Methods: Patients with R/R acute leukemia undergoing allo-HCT at Memorial Sloan Kettering Cancer Center (MSK) between 2009 and 2020 were included. Patients had a marrow blast count of 5% or more at the time of allo-HCT and were classified into one of the following categories: primary induction failure (PIF), at least 2 lines of induction therapy; not in CR (NCR), no more than one line of induction therapy; untreated relapse (UR); and refractory relapse (RR), no remission after at least one line of re-induction therapy for relapse.
Results: We identified 101 patients with acute myeloid leukemia (AML; n=94), acute lymphoblastic leukemia (ALL; n=3), biphenotypic acute leukemia (n=2), and plasmacytoid dendritic cell leukemia (n=2). Table 1 depicts patient and transplant characteristics. Median blast count at the time of allo-HCT was 8% (IQR, 5%,15%). 37 patients had at least one genomic alteration, and 30 patients had circulating blasts at the time of transplant. Grafts were from HLA-identical siblings in 30 patients, from HLA-matched unrelated donors in 51 patients and from HLA-mismatched or HLA-haploidentical donors in 20 patients. 61 and 40 patients received MAC and RIC, respectively. Graft source was unmodified bone marrow (n=8), unmodified peripheral blood stem cells (PBSC; n=80), or CD34+ cellselected peripheral blood stem cells (n=7). Overall survival (OS) at 4 years was 25% (95% CI: 18%, 35%). In univariate analysis, RIC (HR 1.71, p=0.035), presence of circulating blasts (HR 2.21, p=0.002), and high pre-transplant ferritin levels (HR 1.43, p=0.049) were associated with inferior OS. In multivariate analysis, RIC was associated with reduced OS. The 4-year cumulative incidence of relapse/disease progression and non-relapse mortality (NRM) was 54% (95% CI: 43%, 63%) and 24% (CI, 16%, 33%) respectively. In univariate analysis, RIC, presence of circulating blasts, and elevated pre-transplant ferritin levels were associated with increased NRM. In multivariate analysis, RIC and elevated pre-transplant ferritin levels were associated with high NRM. The cumulative incidence of acute graft-vs-host disease (aGVHD) was 45% (95% CI: 35%, 54%) at day 100. There was no statistically significant effect of genomic and molecular parameters on NRM and OS. However, high risk molecular mutations (TP53, RUNX1, ASXL1 or FLT3-ITD) had borderline association with relapse/disease progression (HR 1.7, p=0.073).
Conclusion: Allo-HCT provides durable remission in a select group of patients with relapsed or refractory acute leukemia with an OS of 25% at 4 years. The presence of circulating blasts and elevated ferritin levels at the time of allo-HCT was associated with increased NRM and inferior OS in this retrospective analysis. Known high-risk gene mutations did not affect outcomes in this population. The impact of these parameters should be evaluated in a larger cohort of patients.
Disclosures
Ponce:Seres Therapeutics: Research Funding. Young:Amgen: Other: Stock ownership publicly traded company; Pfizer: Other: Stock ownership publicly traded company; Merck: Other: Stock ownership publicly traded company. Giralt:Novartis: Consultancy; Celgene: Consultancy, Research Funding; Takeda: Consultancy, Research Funding; Jazz Pharmaceutical: Consultancy; Janssen: Consultancy; Spectrum Pharma: Consultancy; Johnson & Johnson: Consultancy, Research Funding; Omeros: Research Funding; Kite: Consultancy; Amgen: Consultancy, Research Funding; Actinuum: Consultancy, Research Funding; Miltenyi: Research Funding. Perales:Sellas Life Sciences: Consultancy; Incyte: Honoraria, Research Funding; Novartis: Honoraria; Kite, a Gilead Company: Honoraria, Research Funding; Miltenyi Biotec: Consultancy, Honoraria; Merck: Consultancy; VectivBio AG: Honoraria; Vor Biopharma: Honoraria; Orca Bio: Consultancy; Karyopharm: Honoraria; Nektar Therapeutics: Consultancy, Honoraria; Abbvie: Honoraria; Cidara Therapeutics: Consultancy; Astellas: Honoraria; Celgene: Honoraria; Bellicum: Honoraria; Servier: Consultancy; DSMB: Other; Omeros: Consultancy; MorphoSys: Consultancy, Honoraria; Takeda: Honoraria; Medigene: Consultancy; Bristol-Mysers Squibb: Honoraria. Shaffer:Gamida Cell: Consultancy; Miltenyi Biotec: Research Funding; Hansa Biopharma: Consultancy. Gyurkocza:Actinium Pharmaceuticals, Inc.: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal